Research Projects
The Spine Biomechanics group comprising engineers from ETH Zurich and surgeons from the Balgrist University Hospital. Together they conduct translational research at the interface between fundamental biomechanical research and the development of clinically applicable methods. The interdisciplinary approach guarantees a high level of expertise required to address relevant clinical issues. The group has established a fully equipped spine lab with custom test setups for biomechanical in-vitro experiments that is on par with the worldwide leading biomechanical spine laboratories. This equipment has been used for a multitude of biomechanical studies on human spine cadavers to investigate and characterize human anatomy and tissue properties. The obtained results and insights are translated into patient-specific, biomechanical models that are used to optimize pre-operative surgical planning.
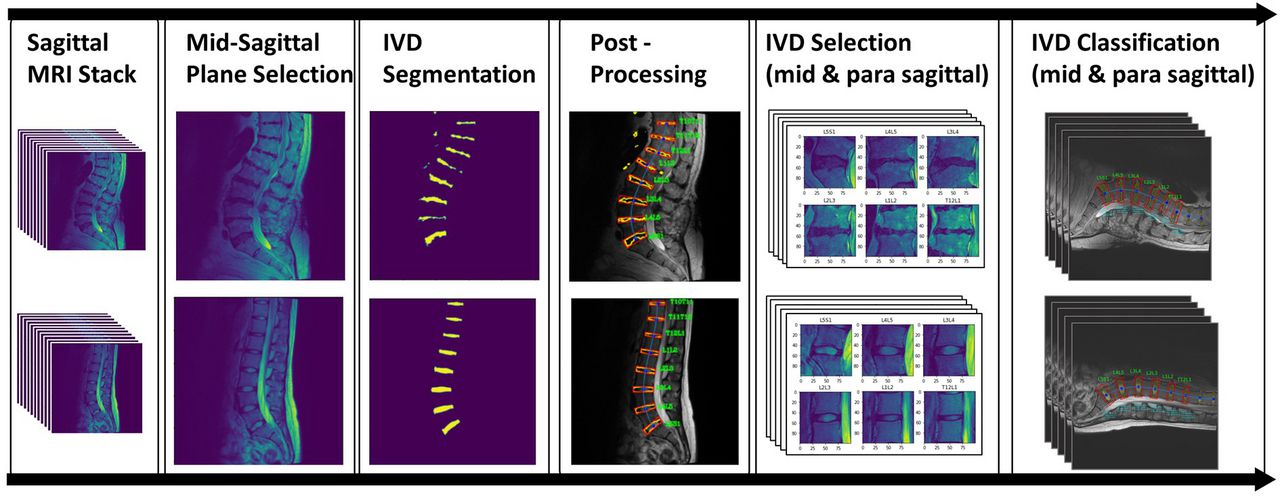
Deep Learning Pipeline for Automatized Assessment of Spinal MRI
The pipeline automatically classifies the degenerative state of lumbar intervertebral discs from raw patients MRI scans.
Low back pain (LBP) is one of the leading causes of work absence and disability worldwide, causing a substantial economic burden to society. LBP is a symptom with many possible causes, comprising a heterogeneous group of patients. Clinical guidelines commonly recommend grouping LBP patients into one of three groups (serious spinal pathology (ca. 1–2%), sciatica/radicular pain (ca. 5%) and non-specific low back pain (>90%)). These entities differ in presentation, prognosis, and management, and the pathoanatomy of the spine must often be visualized for proper diagnosis. The use of magnetic resonance imaging (MRI) is considered the gold standard for the visualization of degenerative and pathological processes in the spine. However, the association between MRI findings and LBP is controversial, and the presence of imaging changes is only weakly associated with pain in the general population. Symptomatic changes are not distinguishable from asymptomatic pathological changes with certainty and the correlation between MRI findings and pain is further complicated by multiple coexisting pathological MRI findings in the same spine or vertebral segment. Despite this, literature on the association between pain and the sum or different patterns of morphological changes is scarce.
In a clinical setting, radiology reports typically consist of a subjectively weighted description of the findings, are qualitative in nature, and subject to substantial intra- and inter-examiner variability. Quantitative reading techniques utilize standard measures or classifications to describe morphological changes within different structures of a vertebral segment. Since manual extraction of relevant quantitative measurements is prohibitively time and labor-intensive, the utilization of machine learning technologies is of great interest as a fast and efficient method to quantitatively assess large amounts of imaging data. This may lead to better recognition of certain patterns that link to a specific clinical presentation, or even aid in predicting outcomes after different interventions.
With the goal of automatizing the whole evaluation process, the pipeline combines multiple task-specific deep learning models into one framework. After training the models on labeled patient data, no further human decision-making or manual annotations are needed during execution. The pipeline includes all steps from MRI input to the segmentation and classification of the intervertebral disc (IVD), offering different radiological gradings of degenerative changes as output (Figure 1).